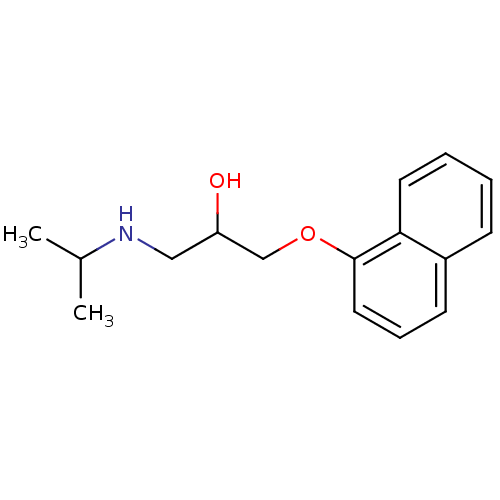
 Avlocardyl PROPRANOLOL,(+) BDBM25761 PROPANOLOL(-) PROPRANOLOL, l- Propanolol,(+/-) CHEMBL27 PROPRANOLOL, d PROPRANOLOL,(-) [2-hydroxy-3-(naphthalen-1-yloxy)propyl](propan-2-yl)amine Anapriline Propanolol Propranolol
Avlocardyl PROPRANOLOL,(+) BDBM25761 PROPANOLOL(-) PROPRANOLOL, l- Propanolol,(+/-) CHEMBL27 PROPRANOLOL, d PROPRANOLOL,(-) [2-hydroxy-3-(naphthalen-1-yloxy)propyl](propan-2-yl)amine Anapriline Propanolol Propranolol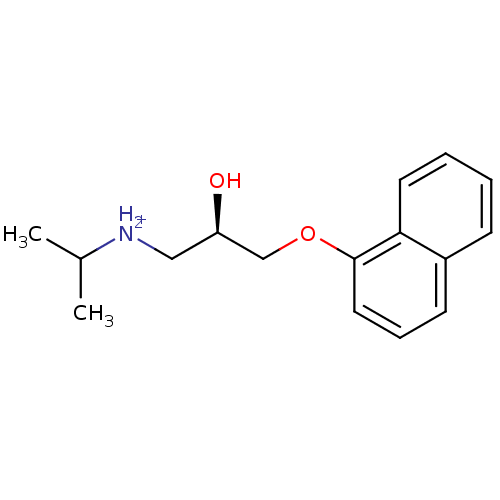 (R)-propranolol BDBM36108
(R)-propranolol BDBM36108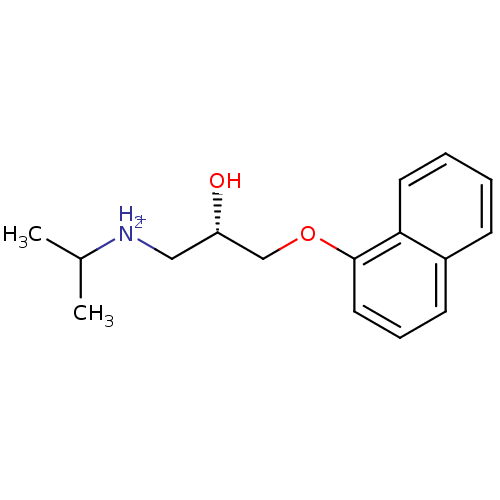 BDBM36109 (S)-propranolol
BDBM36109 (S)-propranolol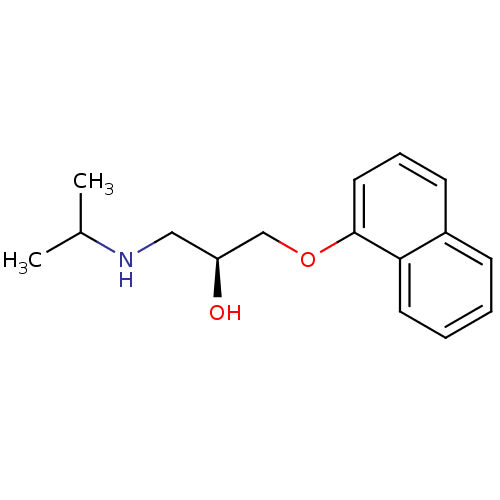 (-)-(S)-Propranolol [2-Hydroxy-3-(naphthalen-1-yloxy)-propyl]-isopropyl-ammonium((-)-propranolol) BDBM50246936 PROPRANOLOL 1-(ISOPROPYLAMINO)-3-(1-NAPHTHYLOXY)-2-PROPANOL CHEMBL452861 S-(-)-propanolol
(-)-(S)-Propranolol [2-Hydroxy-3-(naphthalen-1-yloxy)-propyl]-isopropyl-ammonium((-)-propranolol) BDBM50246936 PROPRANOLOL 1-(ISOPROPYLAMINO)-3-(1-NAPHTHYLOXY)-2-PROPANOL CHEMBL452861 S-(-)-propanolol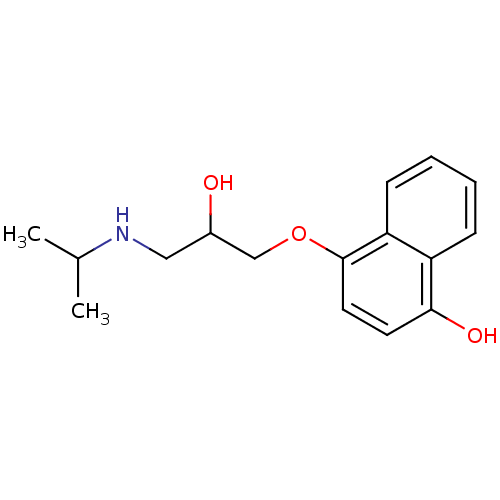 BDBM50405147 4-Hydroxy Propranolol 4-HYDROXYPROPRANOLOL SULFATE
BDBM50405147 4-Hydroxy Propranolol 4-HYDROXYPROPRANOLOL SULFATE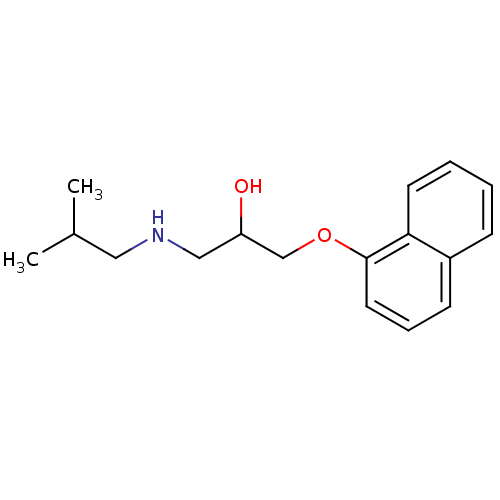 BDBM81886 CHEMBL287651 CAS_3506-09-0 PROPRANOLOL, dl
BDBM81886 CHEMBL287651 CAS_3506-09-0 PROPRANOLOL, dl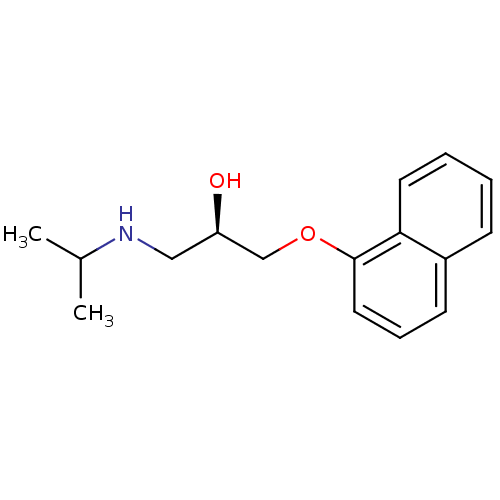 cid_66366 MLS001333595 SMR000875288 (2R)-1-naphthalen-1-yloxy-3-(propan-2-ylamino)propan-2-ol;hydrochloride (2R)-1-(isopropylamino)-3-(1-naphthoxy)propan-2-ol;hydrochloride (2R)-1-(1-naphthalenyloxy)-3-(propan-2-ylamino)-2-propanol;hydrochloride (R)-(+)-Propranolol hydrochloride BDBM60973
cid_66366 MLS001333595 SMR000875288 (2R)-1-naphthalen-1-yloxy-3-(propan-2-ylamino)propan-2-ol;hydrochloride (2R)-1-(isopropylamino)-3-(1-naphthoxy)propan-2-ol;hydrochloride (2R)-1-(1-naphthalenyloxy)-3-(propan-2-ylamino)-2-propanol;hydrochloride (R)-(+)-Propranolol hydrochloride BDBM60973
- Pierson, ME; Lyon, RA; Titeler, M; Schulman, SB; Kowalski, P; Glennon, RA Design and synthesis of propranolol analogues as serotonergic agents. J Med Chem 32: 859-63 (1989)
- Rombouts, FJ; Tovar, F; Austin, N; Tresadern, G; Trabanco, AA Benzazaborinines as Novel Bioisosteric Replacements of Naphthalene: Propranolol as an Example. J Med Chem 58: 9287-95 (2015)
- Nelson, WL; Bartels, MJ; Bednarski, PJ; Zhang, S; Messick, K; Horng, JS; Ruffolo, RR The 3,4-catechol derivative of propranolol, a minor dihydroxylated metabolite. J Med Chem 27: 857-61 (1984)
- Leftheris, K; Goodman, M Synthesis and beta-adrenergic antagonist activity of stereoisomeric practolol and propranolol derivatives. J Med Chem 33: 216-23 (1990)
- Faudone, G; Bischoff-Kont, I; Rachor, L; Willems, S; Zhubi, R; Kaiser, A; Chaikuad, A; Knapp, S; F�rst, R; Heering, J; Merk, D Propranolol Activates the Orphan Nuclear Receptor TLX to Counteract Proliferation and Migration of Glioblastoma Cells. J Med Chem 64: 8727-8738 (2021)
- ChEMBL_684924 (CHEMBL1286339) Displacement of [125I]CYP from rat cerebral cortex 5HT1B receptor in presence of 30 uM propranolol
- ChEMBL_745285 (CHEMBL1775317) Agonist activity at muscarinic M3 receptor in guinea pig trachea assessed as bronchorelaxation activity in presence of propranolol
- ChEMBL_446228 (CHEMBL895333) Agonist activity at adrenergic beta2 receptor in guinea pig tracheal rings assessed as myorelaxing activity on carbachol-induced contraction in presence of propranolol
- ChEMBL_1340540 (CHEMBL3254964) Agonist activity at alpha-adrenergic receptor in Sprague-Dawley rat cerebral cortex assessed as [3H]cAMP accumulation by liquid scintillation spectroscopy in presence of propranolol
- ChEMBL_2216709 (CHEMBL5129841) Invivo antagonist activity at muscarinic M3 receptor activity in Dunkin-Hartley guinea pig trachea assessed as inhibition of carbachol induced contraction in presence of propranolol
- ChEMBL_1904321 (CHEMBL4406543) Agonist activity at human D3R expressed in HEK293 cells assessed as inhibition of forskolin-stimulated cAMP production measured after 5 mins in presence of beta-adrenergic blocker propranolol and coelenterazine H by CAMYEL BRET assay
- ChEMBL_1913985 (CHEMBL4416568) Agonist activity at beta2 adrenergic receptor in guinea pig tracheal smooth muscle assessed as inhibition of histamine-induced tracheal smooth muscle contraction by measuring spasmolysis preincubated for 15 mins in presence of beta adrenergic receptor antagonist propranolol followed by histamine addition
- Inhibition Assay Human recombinant adrenergic β1 receptors expressed in CHO-K1 cells are used in modified Tris-HCl buffer pH 7.4. A 25 aliquot is incubated with 0.03 nM [125I]Cyanopindolol for 120 minutes at 25� C. Non-specific binding is estimated in the presence of 100 μM S(−)-Propranolol. Receptors are filtered and washed, the filters are then counted to determine [125I]Cyanopindolol specifically bound. Compounds are screened at 10 μM.
- Beta2-AR Binding Assay HEK 293 cells stability transfected with cDNA encoding human beta2-AR (provided by Dr. Brian Kobilka, Stanford Medical Center, Palo Alto, Calif.) were grown in Dulbecco's Modified Eagle Medium (DMEM) containing 10% fetal bovine serum (FBS), 0.05% penicillin-streptomycin, and 400 g/ml G418 as previously described (Pauwels et al., Biochem. Pharmacol. 42: 1683-1689, 1991). The cells were scraped from the 150x25 mm plates and centrifuged at 500xg for 5 minutes. The pellet was homogenized in 50 mM Tris-HCl, pH 7.7, with a Polytron, centrifuged at 27,000xg, and resuspended in the same buffer. The latter process was repeated, and the pellet was resuspended in 25 mM Tris-HCl containing 120 mM NaCl, 5.4 mM KCl, 1.8 mM CaCl2, 0.8 mM MgCl2, and 5 mM glucose, pH 7.4. The binding assays contained 0.3 nM [3H]CGP-12177 in a volume of 1.0 ml. Nonspecific binding was determined by 1 uM propranolol.
- Beta-AR binding assay β1-AR binding was done on rat cortical membrane following a previously described procedure (Beer et al., Biochem. Pharmacol. 37: 1145-1151, 1988). In brief, male Sprague-Dawley rats weighing 250-350 g were decapitated and their brains quickly removed. The cerebral cortices were dissected on ice, weighed and promptly transferred to a 50 ml test tube containing approximately 30 ml of 50 mM Tris-HCl, pH 7.8 (at room temperature). The tissues were homogenized with a polytron and centrifuged at 20,000�g for 12 min at 4� C. The pellet was washed again in the same manner and resuspended at a concentration of 20 mg (original wet wt) per 1 ml in the assay buffer (20 mM Tris-HCl, 10 mM MgCl2, 1 mM EDTA, 0.1 mM ascorbic acid at pH 7.8). To block the β2 sites present in the cortical membrane preparation, 30 nM ICI 118-551 was also added to the assay buffer. To wells containing 100 μl of the test drug and 100 μl of [3H]CGP-12177 (1.4 nM final concentration), 0.8 ml of tissue homogenate was added. After 2 hours at 25� C., the incubation was terminated by rapid filtration. Nonspecific binding was determined by 10 μM propranolol.
- Beta1-AR Binding Assay Beta1-AR binding was done on rat cortical membrane following a previously described procedure (Beer et al., Biochem. Pharmacol. 37: 1145-1151, 1988). In brief, male Sprague-Dawley rats weighing 250-350 g were decapitated and their brains quickly removed. The cerebral cortices were dissected on ice, weighed and promptly transferred to a 50 ml test tube containing approximately 30 ml of 50 mM Tris-HCl, pH 7.8 (at room temperature). The tissues were homogenized with a polytron and centrifuged at 20,000xg for 12 min at 4° C. The pellet was washed again in the same manner and resuspended at a concentration of 20 mg (original wet wt) per 1 ml in the assay buffer (20 mM Tris-HCl, 10 mM MgCl2, 1 mM EDTA, 0.1 mM ascorbic acid at pH 7.8). To block the beta2 sites present in the cortical membrane preparation, 30 nM ICI 118-551 was also added to the assay buffer. To wells containing 100 ul of the test drug and 100 ul of [3H]CGP-12177 (1.4 nM final concentration), 0.8 ml of tissue homogenate was added. After 2 hours at 25° C., the incubation was terminated by rapid filtration. Nonspecific binding was determined by 10 uM propranolol.
- Inhibition Assay The CYP2D6 inhibition assays were conducted using Human Liver Microsomes purchased from Invitrogen and designed to screen potential inhibitors of Cytochrome P450 in physiological condition. Initially the following reagents/mixtures were prepared: (i) Assay buffer: 0.1 M Phosphate buffer pH 7.4 (ii) Cofactor: 15 mM stock was prepared in assay buffer. Final concentration in assay 1.5 mM (iii) Substrate 50 mM DMSO stock was prepared for Bufuralol. From this, a 10 mM sub-stock was prepared in MeCN. Further, a working stock solution of 50 μM was prepared in assay buffer. Final concentration in assay 5 μM (iv) Enzyme: 20 mg/mL stock was provided by the manufacturer. Final concentration in assay is 0.25 mg/mL. At the start of the experiment, various concentrations of compound (7 different concs.) or positive control (Quinidine at a single concentration) were prepared in assay buffer. For 1004 of final reaction system, 1.254 of HLM (20 mg/ml), 504 of 2� stock of test compound/reference compound (from each concentration) was added. Subsequently, 104 of substrate (Bufuralol 50 μM) and 104 of Cofactor (NADPH; 15 mM) were added. The volume was increased to 1004 by adding assay buffer. The reaction was then incubated for 10 min at 37� C. After completion of the incubation period, the reaction was terminated by addition of 2004 of chilled MeOH containing internal standard (Propranolol). The samples were than centrifuged and supernatants were analyzed using LCMS/MS. The data normalization was performed with respect to internal standard and % inhibition was calculated with respect to DMSO control.
- Ligand-Displacement Binding Twenty-four hours after adenoviral infection with human β2-AR, HEK293 cells were harvested in lysis buffer, Tris-HCl [5 mM, pH 7.4] containing EGTA [5 mM], and homogenized with 15 strokes on ice. Samples were centrifuged at 30,000�g for 15 minutes to pellet membranes. Membranes were resuspended in binding buffer, Tris-HCl [20 mM, pH 7.4] containing NaCl (120 mM), KCl (5.4 mM), CaCl2 (1.8 mM), MgCl2 (0.8 mM), and glucose (5 mM) and stored in aliquots at −80� C. Binding assays were performed on 5-10 μg of membrane protein using saturating amounts (1-300 pM) of the β-AR-specific ligand [125I]cyanopindolol (ICYP). For competition binding, the 5-10 μg of membrane protein were pretreated with 50 μM of GTPγs (non-hydrolyzable guanosine triphosphate) and then incubated with 125ICYP (50 pM) and different concentrations of fenoterol or its isomers in a total volume of 250 μL. Nonspecific binding was determined in the presence of 20 μM propranolol. Reactions were conducted in 250 μL of binding buffer at 37� C. for 1 hour. The binding reaction was terminated by addition of ice-cold Tris-HCl [10 mM, pH 7.4] to the membrane suspension, followed by rapid vacuum filtration through glass-fiber filters (Whatman GF/C). Each filter was washed three times with an additional 7 mL of ice-cold Tris-HCl [10 mM, pH 7.4]. The radioactivity of the wet filters was determined in a gamma counter. All assays were performed in duplicate, and receptor density was normalized to milligrams of membrane protein. Kd and the maximal number of binding sites (Bmax) for ICYP were determined by Scatchard analysis of saturation binding isotherms.
 Avlocardyl PROPRANOLOL,(+) BDBM25761 PROPANOLOL(-) PROPRANOLOL, l- Propanolol,(+/-) CHEMBL27 PROPRANOLOL, d PROPRANOLOL,(-) [2-hydroxy-3-(naphthalen-1-yloxy)propyl](propan-2-yl)amine Anapriline Propanolol Propranolol
Avlocardyl PROPRANOLOL,(+) BDBM25761 PROPANOLOL(-) PROPRANOLOL, l- Propanolol,(+/-) CHEMBL27 PROPRANOLOL, d PROPRANOLOL,(-) [2-hydroxy-3-(naphthalen-1-yloxy)propyl](propan-2-yl)amine Anapriline Propanolol Propranolol (R)-propranolol BDBM36108
(R)-propranolol BDBM36108 BDBM36109 (S)-propranolol
BDBM36109 (S)-propranolol (-)-(S)-Propranolol [2-Hydroxy-3-(naphthalen-1-yloxy)-propyl]-isopropyl-ammonium((-)-propranolol) BDBM50246936 PROPRANOLOL 1-(ISOPROPYLAMINO)-3-(1-NAPHTHYLOXY)-2-PROPANOL CHEMBL452861 S-(-)-propanolol
(-)-(S)-Propranolol [2-Hydroxy-3-(naphthalen-1-yloxy)-propyl]-isopropyl-ammonium((-)-propranolol) BDBM50246936 PROPRANOLOL 1-(ISOPROPYLAMINO)-3-(1-NAPHTHYLOXY)-2-PROPANOL CHEMBL452861 S-(-)-propanolol BDBM50405147 4-Hydroxy Propranolol 4-HYDROXYPROPRANOLOL SULFATE
BDBM50405147 4-Hydroxy Propranolol 4-HYDROXYPROPRANOLOL SULFATE BDBM81886 CHEMBL287651 CAS_3506-09-0 PROPRANOLOL, dl
BDBM81886 CHEMBL287651 CAS_3506-09-0 PROPRANOLOL, dl cid_66366 MLS001333595 SMR000875288 (2R)-1-naphthalen-1-yloxy-3-(propan-2-ylamino)propan-2-ol;hydrochloride (2R)-1-(isopropylamino)-3-(1-naphthoxy)propan-2-ol;hydrochloride (2R)-1-(1-naphthalenyloxy)-3-(propan-2-ylamino)-2-propanol;hydrochloride (R)-(+)-Propranolol hydrochloride BDBM60973
cid_66366 MLS001333595 SMR000875288 (2R)-1-naphthalen-1-yloxy-3-(propan-2-ylamino)propan-2-ol;hydrochloride (2R)-1-(isopropylamino)-3-(1-naphthoxy)propan-2-ol;hydrochloride (2R)-1-(1-naphthalenyloxy)-3-(propan-2-ylamino)-2-propanol;hydrochloride (R)-(+)-Propranolol hydrochloride BDBM60973